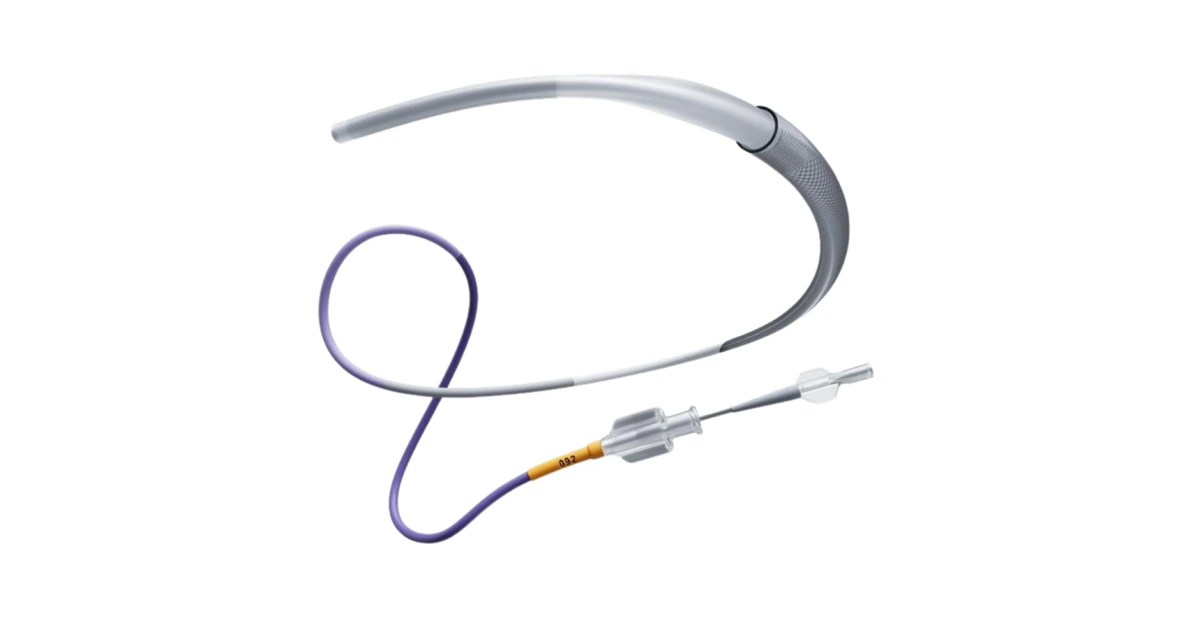
USA – Johnson & Johnson MedTech, a leader in neurovascular care, has launched the CEREGLIDE 92 Catheter System, a next-generation .092” catheter designed to assist in the treatment of acute ischemic strokes.
This new device helps doctors navigate the brain’s complex blood vessels more effectively, improving access to the M1 artery, which is most commonly affected by stroke.
The CEREGLIDE 92 catheter features a larger lumen to support multiple neurovascular tools and reduce blood flow in the M1 artery during procedures.
It also includes INNERGLIDE 9, a special delivery aid that makes it easier to insert and guide interventional devices.
This system is part of Johnson & Johnson MedTech’s expanding stroke treatment portfolio, which includes CEREGLIDE 71, PROWLER EX, and EMBOTRAP III.
Acute ischemic strokes make up 85% of all strokes worldwide, and while many patients benefit from mechanical thrombectomy, some face challenges due to difficulties accessing the affected arteries.
By using the CEREGLIDE 92 catheter, doctors can perform thrombectomy procedures more efficiently by either directly removing blood clots or combining the catheter with a stent retriever for better blood flow restoration.
Dr. Brian Jankowitz, Chief of Neurosurgery and Co-Chair of the Stroke and Neurovascular Program at Hackensack Meridian Neuroscience Institute, emphasized the importance of this innovation. He stated,
“Vessel tortuosity and other anatomical challenges are present in approximately 50 percent of mechanical thrombectomy cases, which can increase procedure time or prevent ability to treat. As an early user of CEREGLIDE 92 and INNERGLIDE 9, this super large-bore catheter system helps provide quick and seamless access to the M1 with the benefit of flow control, allowing us to rapidly reperfuse the patient and extend the benefits of next-generation stroke technology to more patients.”
The CEREGLIDE 92 catheter incorporates advanced features such as BRITE-LINE Technology, ensuring full visibility under fluoroscopy, and TruCourse Technology, which enhances flexibility and trackability, allowing smoother navigation through blood vessels.
Mark Dickinson, Worldwide President of Neurovascular at Johnson & Johnson MedTech, highlighted the company’s commitment to innovation:
“Johnson & Johnson MedTech is continuing to innovate to help address the access challenges physicians face during mechanical thrombectomy procedures with the launch of CEREGLIDE™ 92. This new catheter system is designed to assist physicians in treating their patients and maximizing procedural outcomes, to ultimately change the trajectory of stroke.”
In line with sustainability efforts, Johnson & Johnson MedTech will provide electronic instructions for use (e-IFUs) for the CEREGLIDE 92 system.
This approach aims to reduce the company’s environmental footprint, lower CO2 emissions from shipping, and minimize medical waste disposal in healthcare systems.
The CEREGLIDE 92 Catheter System is now available for commercial use in the United States.
XRP HEALTHCARE L.L.C | License Number: 2312867.01 | Dubai | © Copyright 2025 | All Rights Reserved