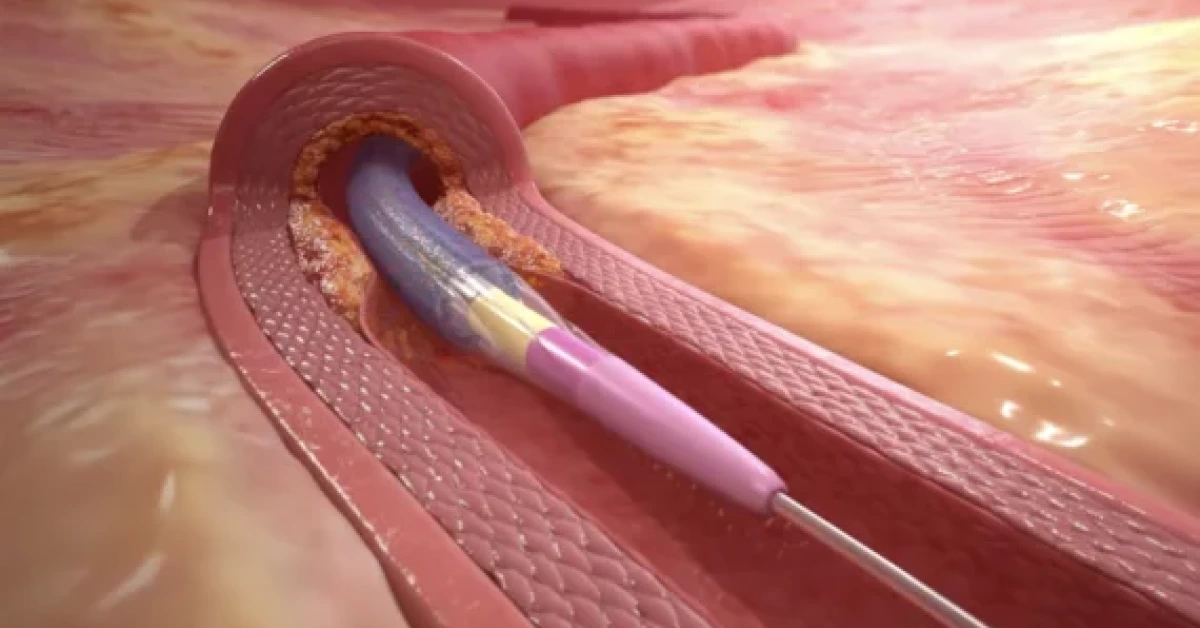
IRELAND – Medtronic has begun a global trial to evaluate its Prevail paclitaxel-coated balloon catheter in treating in-stent restenosis (ISR) and small vessel disease in patients with coronary artery disease (CAD).
The first patient has been enrolled, marking a key milestone in the study. The Prevail drug-coated balloon (DCB) is designed to inflate within the artery during a percutaneous coronary intervention (PCI), releasing paclitaxel to prevent ISR recurrence.
This reduces the need for repeat procedures. The balloon’s FreePac coating has already shown effectiveness in treating both CAD and peripheral artery disease.
Dr. Ziad Ali, director of the DeMatteis Cardiovascular Institute at St. Francis Hospital and Heart Center in New York, performed the first procedure in this global study.
He emphasized the importance of innovative solutions for CAD, a condition affecting over 315 million people worldwide.
“This trial will not only bring the use of this innovative DCB technology to patients with previously treated blockages—where stents have failed—but also to new blockages in small vessels, where stents may not perform as well in coronary arteries,” Ali stated.
The Prevail Global study aims to enroll up to 1,205 patients across the United States, Europe, and Asia.
The primary goal is to assess the safety and effectiveness of the Prevail DCB, with the one-year target lesion failure rate as the key measure.
Medtronic’s Prevail DCB has had CE mark approval since 2001 and is already available in multiple countries.
However, Medtronic hopes data from this study will support regulatory approvals in the United States and Japan.
Drug-coated balloons gaining momentum in the U.S.
While DCBs have been widely used worldwide, they are only now gaining traction in the U.S. Boston Scientific’s Agent DCB became the first FDA-approved coronary DCB in March 2024, marking a significant moment for interventional cardiologists.
Medtronic’s trial could further expand DCB use in coronary procedures, offering new treatment options for CAD patients.
XRP HEALTHCARE L.L.C | License Number: 2312867.01 | Dubai | © Copyright 2025 | All Rights Reserved